What Is Digital Pathology? Core Definition & Components
Think of digital pathology as the evolution from film cameras to digital photography, but for medical diagnosis. Instead of pathologists examining tissue samples through a microscope eyepiece at a glass slide, they can now analyze the same samples on high-resolution computer screens—with capabilities that far exceed what traditional microscopy could offer.
Here’s the process: specialized scanners capture high-resolution digital images of glass slides containing tissue samples, creating what are called whole slide images (WSIs). These digital slides can then be viewed, shared, measured, and analyzed on any computer system, enabling instant collaboration between medical experts worldwide. It’s like Google Maps for tissue samples: zoom in, annotate, measure, and share instantly.
According to the Digital Pathology Association, this technology creates a dynamic, image-based environment that enables the acquisition, management, and interpretation of pathological information in ways that were impossible with conventional microscopy. The transformation opens up new possibilities for faster diagnosis, AI-assisted analysis, and global access to specialized expertise.
Key Components That Make Digital Pathology Work
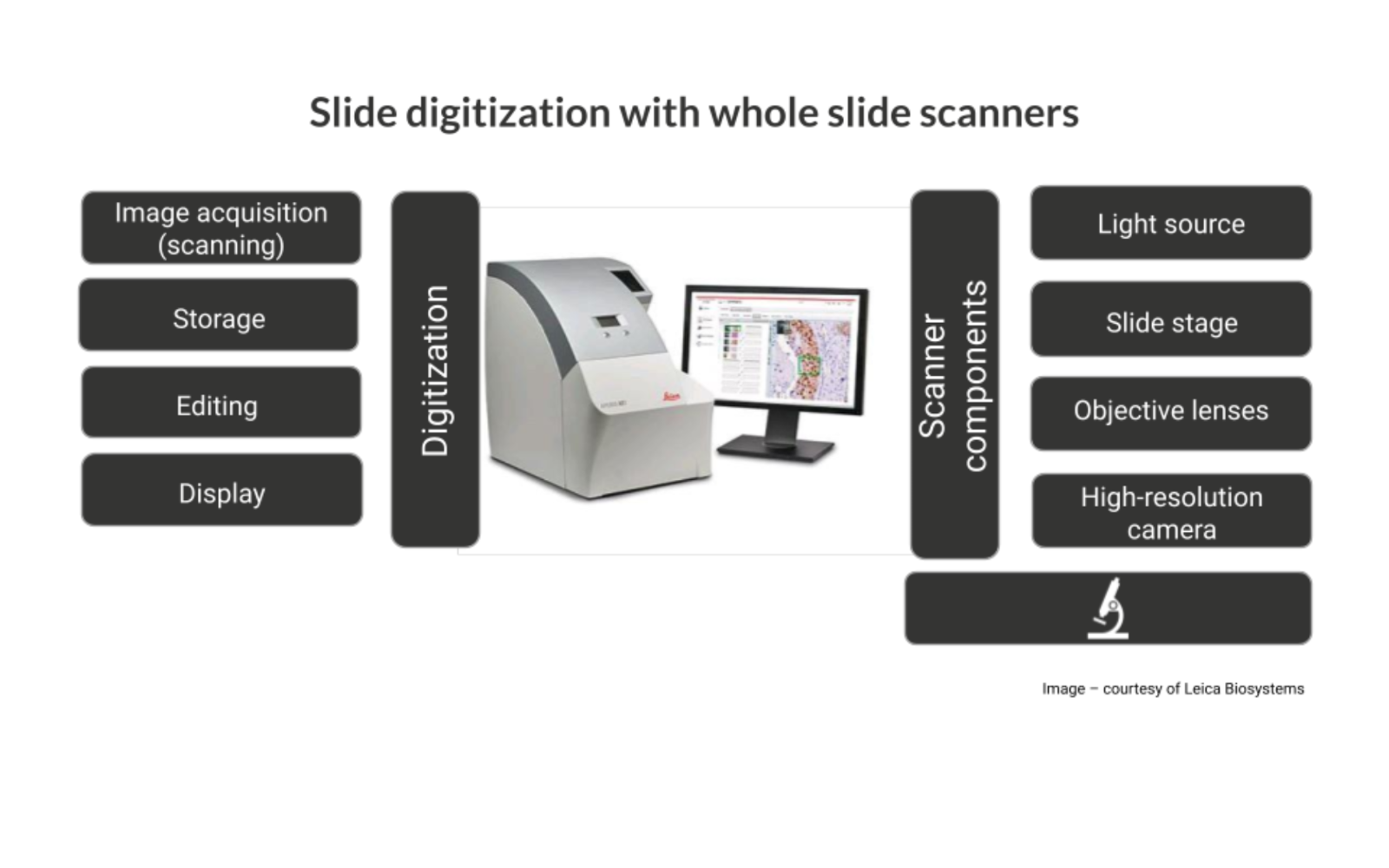
Whole Slide Imaging (WSI) Scanners: The first scanners were clunky instruments that took up large footprints – with long processing times to digitize just a few slides at a time (up to 6 minutes/slide), expensive storage, and limited use cases. Scanners today can scan slides in as little as 30 seconds, be configured for multiple magnifications, and handle up to 1,000 slides simultaneously.
According to the American Journal of Clinical Pathology, as of November 2024, six whole slide imaging systems with unique scanners have received FDA 510(k) clearance for primary diagnostic use. These include the Philips IntelliSite Pathology Solution (first approved in 2017, with version 5.1 in 2024), Leica Biosystems Aperio AT2 DX (2019) and GT 450 DX (2024), Hamamatsu Photonics NanoZoomer S360MD (2022), and most recently, the Roche Digital Pathology Dx system featuring the VENTANA DP 200 scanner (June 2024) and DP 600 scanner (December 2024).
Recent clinical performance studies demonstrate that these FDA-cleared systems show remarkably consistent results, with digital reads typically achieving 92-96% diagnostic accuracy compared to manual microscopy.
Digital Slide Management Software: These platforms enable pathologists to view, annotate, compare, and share digital images. Modern systems integrate seamlessly with scanners, Laboratory Information Systems (LIS), and image analysis software to maintain complete diagnostic workflows.
High-Performance Storage Systems: Digital pathology generates substantial data volumes that require specialized storage infrastructure. Whole slide images typically range from 100MB to 2GB per slide, depending on scan resolution, tissue size, and compression settings. A single slide scanned at 40x magnification can generate data equivalent to two hours of HD video.
Organizations typically implement tiered storage architectures combining high-speed local storage for active cases with archival systems for long-term retention. While cloud-based solutions offer scalability advantages, many healthcare institutions maintain on-premise storage to address data sovereignty requirements and ensure compliance with regulations like HIPAA.
AI-Powered Analysis Tools: Digital pathology AI has evolved from basic computer vision applications to sophisticated multimodal AI systems. While early tools focused on image analysis for automated cell counting and pattern recognition, today’s platforms integrate large language models that can leverage information from pathology reports and LIS.
Modern AI-assisted workflows now encompass cancer detection algorithms, intelligent case prioritization based on urgency and complexity, automated electronic health record (EHR) queries, and AI-powered report generation using voice recognition technology. This evolution transforms AI from a simple diagnostic aid into a comprehensive workflow optimization platform.
“Digital Pathology Ecosystem” showing the interconnected components: Glass slide → Scanner → Storage (e.g. in the cloud) → Software → AI → Pathologist]
How Digital Pathology Works: The Technical Workflow
Digital pathology transforms traditional microscope-based diagnosis through an integrated workflow that combines traditional tissue processing with advanced digital technologies. Here’s the complete step-by-step process:
Step 1: Traditional Tissue Preparation
The process begins identically to conventional pathology: tissue collection through biopsy or surgery, fixation in formalin, paraffin embedding, sectioning, and staining. This foundation remains unchanged because it provides the cellular detail pathologists need for accurate diagnosis.
Step 2: Pre-Scanning Preparation
Before digitization, slides must be prepared for scanning. This involves cleaning slides to remove dust particles and debris, then loading them into the scanner. According to current workflow analysis, these manual steps are increasingly being automated—pre-scanning cleaning can be replaced by during-scanning or post-scanning image analysis algorithms that detect artifacts, trigger rescanning, or alert scanning technicians about specific problems, while robotic loading systems can automate slide handling.
Step 3: High-Resolution Digital Scanning
WSI scanners capture tissue sections using either tile-based or line-based scanning methods. Both technologies use integrated cameras and motorized stages to systematically image the entire tissue area, with tiles capturing square field-of-view sections while line scanners create continuous stripe images. These individual captures are then computationally stitched together to generate a seamless, zoomable digital representation that recreates the traditional microscope navigation experience.
Step 4: Image Management and Storage
Once digitized, images are usually automatically named and placed into management systems where pathologists and researchers can access them with appropriate permissions. Digital slides are maintained in image management systems (IMS) that allow for archival and intelligent retrieval. This step includes integration with Laboratory Information Systems (LIS) to maintain complete diagnostic workflows.
Step 5: Image Analysis (Added Value Step)
Image analysis represents the primary added value of digital pathology—capabilities impossible with glass slides alone. Modern image analysis can predict molecular markers from H&E slides, predict patient survival, and replace certain IHC markers through computational analysis. Deep learning advances in computer vision have revolutionized this field, allowing the extraction of clinically and scientifically important information previously undetectable by human observers.
Step 6: Digital Diagnosis and Reporting
Once images are stored in the image management system, pathologists access them through integrated or standalone viewing software. According to current digital pathology workflows, viewing capabilities are embedded throughout the process—scanners typically include viewing software for scan planning and quality assessment, while image management systems often incorporate their own viewing platforms. Pathologists use these digital viewing tools on high-resolution monitors to make diagnoses, navigating the zoomable images much like they would adjust microscope magnification. Following diagnosis, pathologists generate reports that document their findings and may include results from automated image analysis, creating comprehensive diagnostic documentation that integrates both traditional morphological assessment and computational insights.
Step 7: Archiving and Retrieval
Unlike physical glass slides stored in basements, digital images require sophisticated archiving solutions. Organizations must establish systems for long-term storage (local or cloud-based) and retrieval, including preservation of metadata embedded in image files and links to associated reports and other imaging studies.
According to industry analysis, typical digital pathology practices require at least a petabyte of storage per year, with high-resolution whole slide images potentially exceeding 1GB each. To address these massive scale requirements, specialized medical data archiving companies offer comprehensive digital pathology storage solutions featuring multi-tiered cloud storage using industry-standard S3 protocols, with immediate access from Active/Hot tiers, minute-to-hour retrieval from Warm tiers, and long-term Cold tier archival for compliance requirements.
These enterprise solutions maintain chain of custody documentation, store both physical slides in temperature-monitored facilities meeting College of American Pathologists requirements and their corresponding digital images in secure cloud environments. Advanced archival systems also enable AI training datasets and data monetization opportunities through anonymized slide libraries, transforming archived pathology assets from storage costs into potential revenue streams for research collaboration and educational purposes.
Step 8: Integrated Quality Control
Once images are stored in the image management system, pathologists access them through integrated or standalone viewing software. According to current digital pathology workflows, viewing capabilities are embedded throughout the process—scanners typically include viewing software for scan planning and quality assessment, while image management systems often incorporate their own viewing platforms. Pathologists use these digital viewing tools on high-resolution monitors to make diagnoses, navigating the zoomable images much like they would adjust microscope magnification. Following diagnosis, pathologists generate reports that document their findings and may include results from automated image analysis, creating comprehensive diagnostic documentation that integrates both traditional morphological assessment and computational insights.
Step 6: Digital Diagnosis and Reporting
Quality control occurs throughout the entire digital pathology workflow, not just as a final step. These quality control components must be integrated at specific stages:
Pre-Scanning Quality Control:
- High-quality slide preparation: Ensuring slides are thin, fold-free, and suitable for single-focus scanning, as digital pathology is “a lot less permissive to artifacts” than traditional microscopy
- Tissue labeling verification: Confirming that the correct labels match the correct specimens before digitization
System-Level Quality Control:
- Scanner calibration: Validating that scanning parameters produce diagnostic-quality images and that colors are represented adequately
- Digital pathology system validation: Compliance with regulatory frameworks and validation of machines, software, and processes throughout the pipeline
Post-Scanning Quality Control:
- Image quality assessment: Verifying that digitized images contain sufficient tissue, ideally all tissue is in focus, and adequate diagnostic material exists for pathologists to make diagnoses or researchers to answer their research questions
Ongoing Quality Control:
- Image analysis quality control: Monitoring algorithmic results for accuracy when computational tools are employed
Currently, many quality control steps traditionally performed manually can now be automated through specialized algorithms that detect artifacts and can trigger rescans when image quality is insufficient for diagnostic purposes.
Why Digital Pathology Matters: Faster, More Accurate Diagnoses
For patients awaiting critical diagnoses, every hour matters. Digital pathology addresses the fundamental bottlenecks that slow traditional pathology workflows.
Speed: Eliminating Physical Transport Delays
With digital pathology, diagnosis no longer needs to be delayed by the physical shipment of tissue samples or the wait times that result when pathologists are out of the office.
Traditional consultations requiring expert opinions can take days or weeks as slides are physically transported between institutions. Digital pathology enables instant sharing across networks, reducing consultation times from days to hours.
Accessibility: Bridging Geographic Gaps
Digital pathology enables remote diagnostics, allowing pathologists to provide expert opinions regardless of location. This is particularly valuable for:
- Rural hospitals lacking specialized pathologists
- Developing countries with limited pathology infrastructure
- Emergencies requiring immediate expert consultation
Enhanced Collaboration
Multiple pathologists can simultaneously review the same case, facilitating real-time collaboration for complex diagnoses. This peer review capability improves diagnostic accuracy and provides valuable learning opportunities. Beyond clinical applications, digital pathology enables unprecedented research collaboration opportunities. Digital slide libraries can be shared globally to support AI algorithm development, with researchers accessing anonymized datasets to train machine learning models for pattern recognition and diagnostic assistance. Pharmaceutical companies and research institutions can collaborate more effectively on drug development studies by sharing digitized tissue samples and corresponding analytical results across geographic boundaries, accelerating biomarker discovery and therapeutic research timelines.
Improved Data Management
Digital pathology transforms data management from a logistical challenge into a strategic advantage, particularly critical for clinical trials. Unlike glass slides that can break, fade, or be misplaced, digital slides provide retrievable archives that maintain image quality as time passes, with complete audit trails and chain of custody documentation.
In clinical trials, this enhanced data management capability enables real-time sharing of pathology data across multiple sites and regulatory jurisdictions while maintaining compliance with Good Clinical Practice (GCP) guidelines. Digital pathology systems can automatically integrate with electronic data capture (EDC) systems, linking tissue-based endpoints to patient outcomes and enabling faster regulatory review processes. The ability to retrospectively analyze archived digital slides allows for biomarker validation studies and post-hoc analysis that would be a lot more difficult or impossible whit glass slides.
Digital pathology also revolutionizes biobank operations by linking digital images directly to stored tissue blocks, enabling researchers to select samples based on actual tissue content rather than relying solely on metadata descriptions. This visual verification capability allows scientists to identify specific tissue architectures, cell populations, or pathological features online before requesting physical samples, dramatically improving research efficiency and reducing unnecessary sample consumption. Biobank users can browse digital slide libraries to locate precisely the tissue characteristics needed for their studies, transforming sample selection from a metadata-dependent process into a content-verified workflow.
The Game-Changing Role of AI in Digital Pathology
Artificial intelligence is transforming digital pathology from a digitization tool into an intelligent diagnostic and research platform. The combination creates unprecedented capabilities for pattern recognition and quantitative analysis, as well as for the combination of different data modalities in multimodal applications.
Current AI Applications in Digital Pathology
Digital pathology AI has evolved from basic image analysis to encompass three distinct categories: image analysis, natural language processing, and multimodal combinations of both approaches.
Image Analysis Applications: AI transforms routine quantitative tasks by automating cell counting and measurement—eliminating tedious manual processes like counting Ki-67 positive nuclei across multiple fields of view. These algorithms work on both whole slide images and static microscope camera images, providing consistent, objective measurements of cellular features.
Computer-aided diagnostics represents a breakthrough application, exemplified by the first FDA-cleared AI tool for pathology diagnosis. According to validation studies, pathologists using AI assistance achieved seven percentage points better accuracy than those working alone, with the combined pathologist-AI team outperforming both individual pathologists and AI algorithms independently.
Molecular Prediction Capabilities: Advanced AI models predict molecular properties directly from tissue images by training on matched image-molecular datasets. These systems can determine mutation status from H&E images alone—a capability already deployed clinically. According to a case report by Dr. Barliz Waissengrin, an AI molecular analysis tool enabled rapid EGFR mutation prediction in lung tissue, allowing faster PCR confirmation instead of next-generation sequencing, accelerating treatment decisions while reducing diagnostic costs.
Virtual staining represents another molecular prediction application, where AI generates immunohistochemistry and special stain predictions from standard H&E images, effectively predicting chemical and molecular tissue properties computationally.
Natural Language Processing Applications: Large language models now address pathology’s text-heavy workflows through speech-to-text systems trained on pathologist vocabulary and dictation patterns, automated structured report generation from verbal case descriptions, and intelligent search capabilities that answer questions about institutional databases using retrieval-augmented generation (RAG) technology to control AI hallucinations.
Multimodal Integration: Leading pharmaceutical companies are combining image analysis AI with language model capabilities to accelerate drug development, representing the convergence of visual and textual AI applications in competitive research environments.
The Future: Predictive and Personalized Medicine
Pathology images contain prognostic data that specialized software can quantify to predict tumor aggressiveness and patient outcomes—insights often invisible even under microscopic examination.
The first FDA-cleared multimodal AI tool for prostate cancer demonstrates this capability clinically. According to regulatory filings, the system analyzes digital pathology images and clinical data to predict the 10-year risk of distant metastasis and cancer-specific mortality. Validated through phase 3 trials, it identifies which patients benefit from hormone therapy and respond optimally to targeted treatments like abiraterone.
These AI-powered insights enable:
- Personalized treatment selection based on individual tissue characteristics rather than population statistics
- Prognostic assessments providing concrete 10-year outcome data for patient counseling
- Targeted therapy optimization with results available within 2-3 days of specimen receipt
This represents a shift from reactive to predictive medicine, where AI transforms routine pathology slides into comprehensive prognostic tools enabling personalized cancer care at the point of diagnosis.
Beyond Glass Slides: The Future of Glassless Pathology
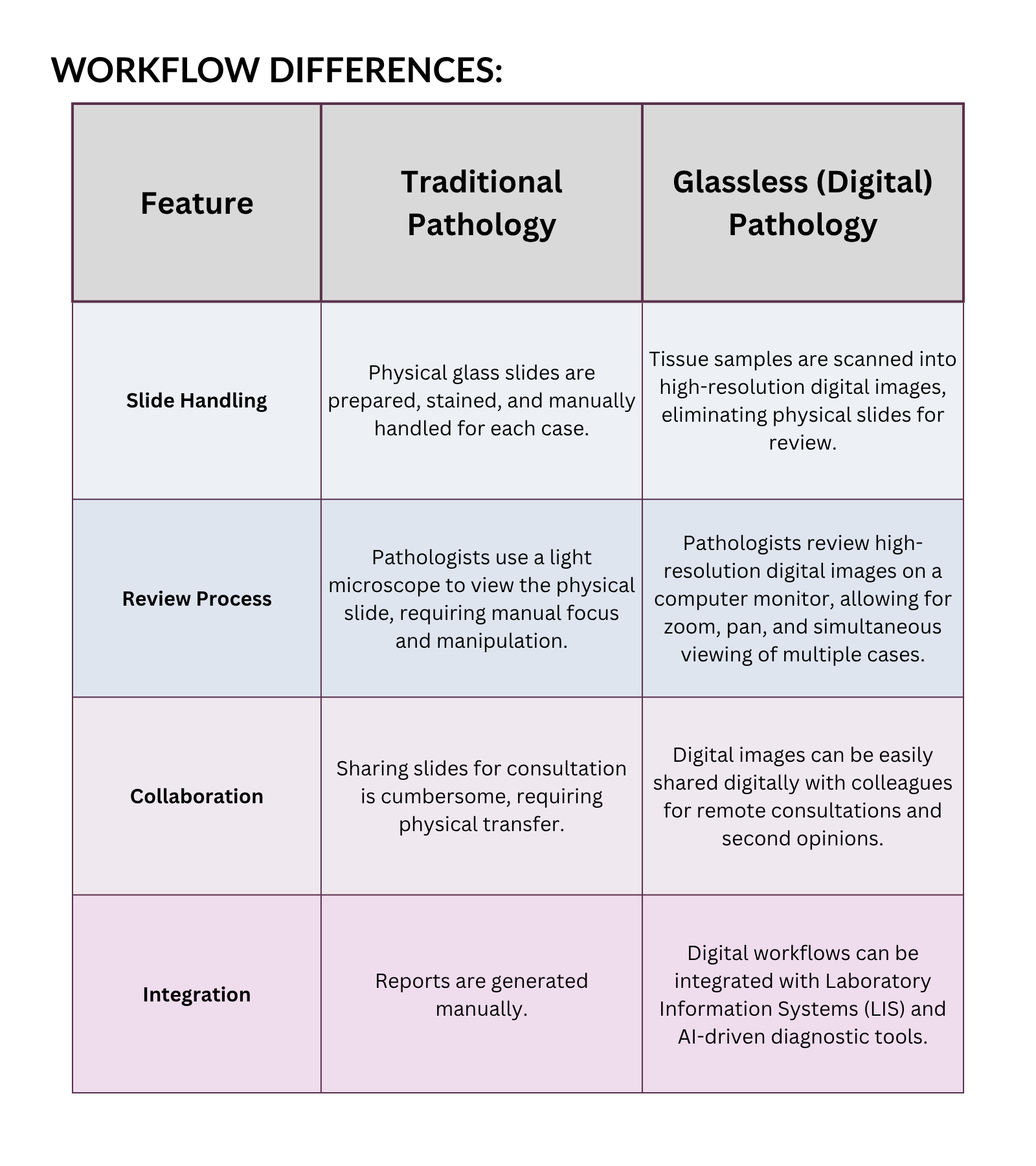
Traditional digital pathology still depends on preparing glass slides, which is time-consuming and requires specialized laboratory infrastructure. New imaging approaches are changing this paradigm by enabling the creation of diagnostic-quality digital images directly from intact tissue, without the intermediate step of slide preparation.
Direct-to-Digital Imaging from Tissue Surface: Microscopy with ultraviolet surface excitation (MUSE) is advancing histology by providing a rapid, non-destructive method to visualize tissue in minutes. By targeting only the superficial layers of a specimen, this approach reveals detailed structural information without the need for thin sectioning.
How Glassless Pathology Works: Instead of cutting and staining slides, glassless pathology uses a straightforward workflow:
- Direct Tissue Imaging: Fresh or fixed samples can be placed as-is into the imaging setup.
- UV Excitation: Short-wavelength light (around 275–280 nm) excites intrinsic and dye-enhanced signals that are captured as high-resolution images.
- Virtual Staining: Computational methods translate these signals into H&E-like views familiar to pathologists.
- Instant Results: Images suitable for review are available in minutes, bypassing lengthy histology processing.
Advantages of Glassless Pathology
- Speed: Delivers high-quality images within minutes, accelerating imaging workflows and reducing turnaround time.
- Safety: Minimizes or removes the need for harsh chemicals traditionally required for tissue processing.
- Molecular Preservation: Gentle preparation preserves nucleic acids and proteins for downstream genomic and molecular assays.
Accessibility: Simplifies pathology imaging, making advanced diagnostics possible even in settings with limited histology infrastructure.
Other Emerging Glassless Technologies
FIBI (Fluorescence-Imitating Brightfield Imaging)
FIBI leverages blue light to generate intrinsic color patterns reminiscent of H&E staining. Both MUSE and FIBI facilitate direct on-site imaging without frozen section processing. Early validation studies show promising results: according to research by Dr. Richard Levenson’s team, validation achieved a 97% concordance between glassless FIBI and standard pathologist H&E reads—a remarkable result suggesting clinical utility.
Additional Direct Imaging Methods
- Light-sheet microscopy for 3D tissue visualization
- Optical coherence tomography (OCT) for real-time imaging
- Multiphoton microscopy for enhanced contrast without traditional staining
- Fluorescence confocal microscopy for rapid examination of fresh specimens
- Photoacoustic microscopy for functional tissue assessment
- Optical sectioning microscopy for layer-by-layer visualization of intact tissue without physical slicing
“Traditional vs Glassless Pathology: Side-by-Side Comparison” showing workflow differences, time savings, and advantages”
Implementation Challenges and Solutions
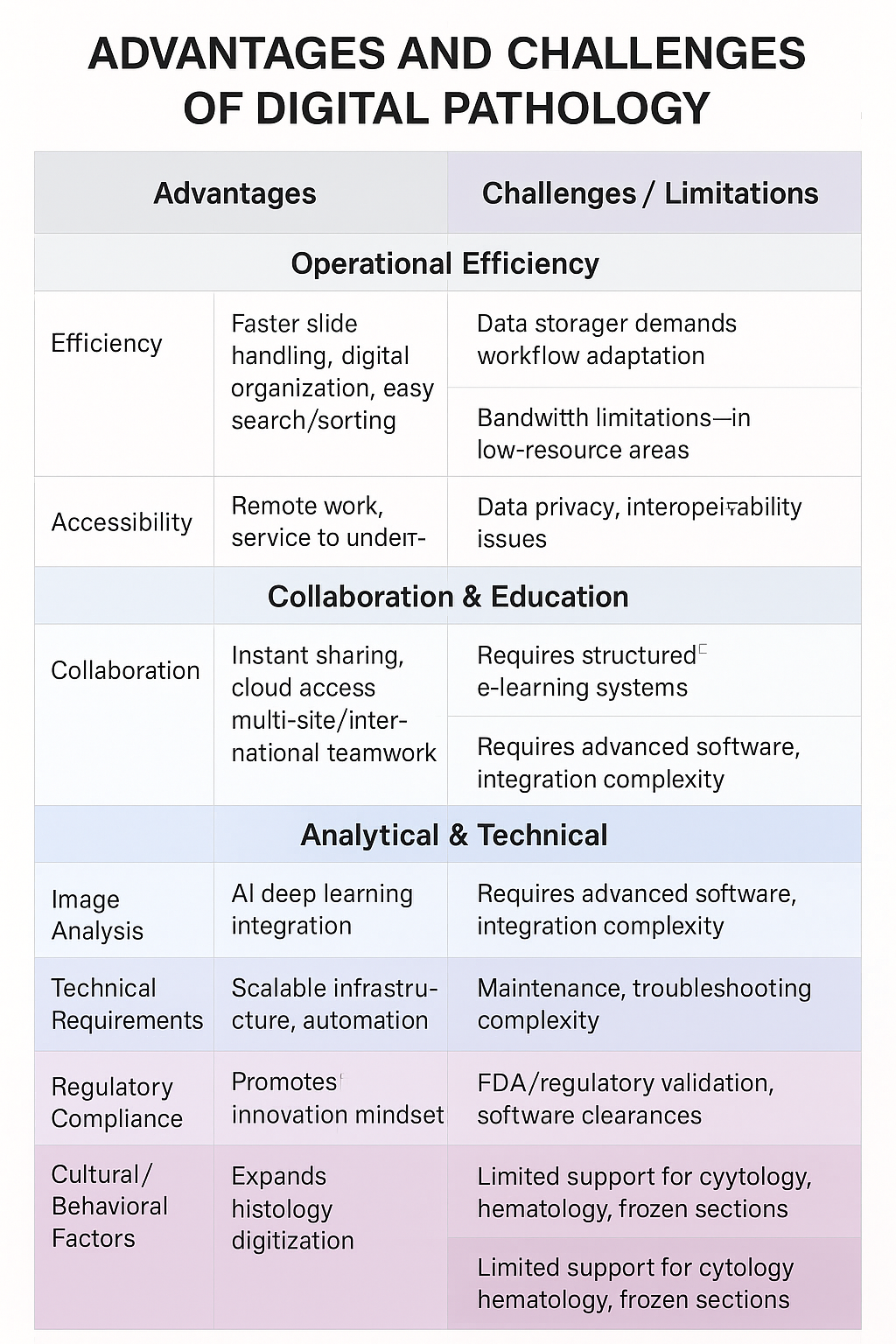
Despite its transformative potential, digital pathology adoption faces significant hurdles that healthcare and research organizations must navigate carefully. These challenges are not only technical and regulatory but also cultural and psychological, affecting how quickly institutions can realize the benefits.
Technical and Infrastructure Challenges
High Initial Investment: Implementing digital pathology involves more than buying a scanner. Costs extend to image management systems, integration with existing IT infrastructure, licensing fees for image analysis tools, and ongoing software maintenance. While this equipment may seem less expensive than other medical technologies (like MRI), the return on investment is not immediate, and organizations must budget for recurring costs.
Storage and Bandwidth Requirements: Digital pathology generates massive amounts of image data, demanding robust IT infrastructure for storage, backup, and smooth network performance. Without proper systems, even simple tasks like uploading or retrieving slides can become bottlenecks.
Technical Troubleshooting: A defining feature of digital pathology adoption is the need for constant troubleshooting. Reliable scanners, storage solutions, and network connections are all essential, but interoperability across systems remains uneven. Encouragingly, vendors are beginning to collaborate by offering integrated workflows where scanners, image management systems, and analysis tools are pre-validated to work together.
Regulatory and Validation Requirements
Compliance and Validation: Every component—scanner, software, archiving system—requires validation before clinical use or use in a regulated environment. This process can disrupt daily workflows, requiring dedicated validation teams and a strong institutional commitment. U.S. FDA oversight and CAP guidelines add further complexity, including washout periods and slide-to-digital concordance studies.
Human Factors and Training
Resistance to Change: Perhaps the greatest hurdle is cultural rather than technical. Even though digital pathology makes life easier—cleaner workspaces, searchable slides, and instant collaboration—adoption in medicine is slower than in consumer technology. Hierarchical structures and long-standing practices in healthcare can create resistance, especially among experienced professionals who may feel threatened by new tools. Managing this change requires sensitivity to interpersonal dynamics and active engagement of leadership, pathologists, technicians, and administrators.
Digital Literacy and Workflow Adaptation: Pathologists and histotechnologists must adapt to new digital workflows. This includes navigating large slide archives digitally, learning new ergonomics for screen-based work, and understanding image analysis outputs. Training is critical to ensure confidence and maintain diagnostic accuracy.
Color Calibration and Standardization: Consistent color reproduction remains essential for reliable diagnosis. Standardized calibration protocols across monitors and systems are required to ensure reproducibility.
Practical Solutions for Successful Implementation
- Phased Rollout Strategy: Begin with consultations, education, and research before scaling to primary diagnosis.
- Hybrid Workflows: Maintain glass slide capabilities during the transition to avoid disruptions.
- Collaborative Planning: Involve pathologists, IT teams, laboratory managers, and administrators to align expectations and smooth implementation.
- Targeted Tools for Difficult Subspecialties: Cytology, hematology, and frozen sections remain harder to digitize due to specimen variability. Specialized tools are emerging to address these use cases, but organizations should plan around current limitations.
The Future of Digital Pathology
Digital pathology continues evolving rapidly, driven by technological advances and changing healthcare needs.
Integration with Other Diagnostic Modalities
Multi-modal Diagnostics: Future platforms will integrate pathology images with radiology, genomics, and clinical data for comprehensive diagnostic assessments.
Molecular Pathology Integration: Digital platforms will seamlessly incorporate molecular testing results, creating unified diagnostic reports that combine morphologic and molecular findings.
Advanced AI and Machine Learning
Predictive Analytics: AI systems will predict patient outcomes, treatment responses, and disease progression based on tissue characteristics invisible to human observation.
Real-time Decision Support: Intelligent systems will provide real-time diagnostic suggestions and alerts during pathologist review.
Global Accessibility and Standardization
Standardized Protocols: In response to the proliferation of proprietary formats and the growing demand for large-scale, multi-center, and AI-driven digital pathology solutions, the community has advanced a set of interoperable image formats engineered for both long-term preservation and high-performance analysis.
Telepathology Expansion: Digital pathology will enable pathology services in underserved regions globally, democratizing access to expert diagnosis.
Regulatory Evolution
As established earlier in this article, six whole slide imaging systems with unique scanners have received FDA 510(k) clearance for primary diagnostic use as of November 2024, making scanner approval a standard regulatory pathway. With multiple whole slide scanners being cleared as medical devices, current regulatory focus centers on AI algorithm validation and Software as Medical Device (SaMD) frameworks for digital pathology applications. According to FDA guidance, AI tools require clinical validation studies demonstrating non-inferiority to traditional diagnostic methods before receiving clearance for primary diagnosis.
Regulatory frameworks will continue evolving to address AI validation, data privacy, and international standardization.
Conclusion: Embracing the Digital Transformation
Digital pathology represents more than technological advancement—it’s a fundamental shift in how we approach disease diagnosis and patient care. From eliminating geographic barriers to enabling AI-powered insights, digital pathology is transforming every aspect of pathology practice.
The emergence of glassless pathology technologies like MUSE represents the next evolutionary step, promising even faster, safer, and more accessible diagnostic capabilities.
For healthcare organizations, the question isn’t whether to adopt digital pathology—it’s how quickly and effectively they can implement these transformative technologies.
The patients depending on accurate, timely diagnoses, deserve nothing less than the full potential of digital pathology. As we continue advancing this technology, we move closer to a future where geographic location, resource limitations, and human error no longer stand between patients and the expert diagnoses they need.
The future of pathology is digital, intelligent, and accessible to all.













Comments are closed.