If you joined me live at 6 a.m. Pennsylvania time for this week’s DigiPath Digest, you already know we started with some classic live-stream chaos—muted microphones, screens that didn’t want to share, and Facebook comments disappearing into the digital void.
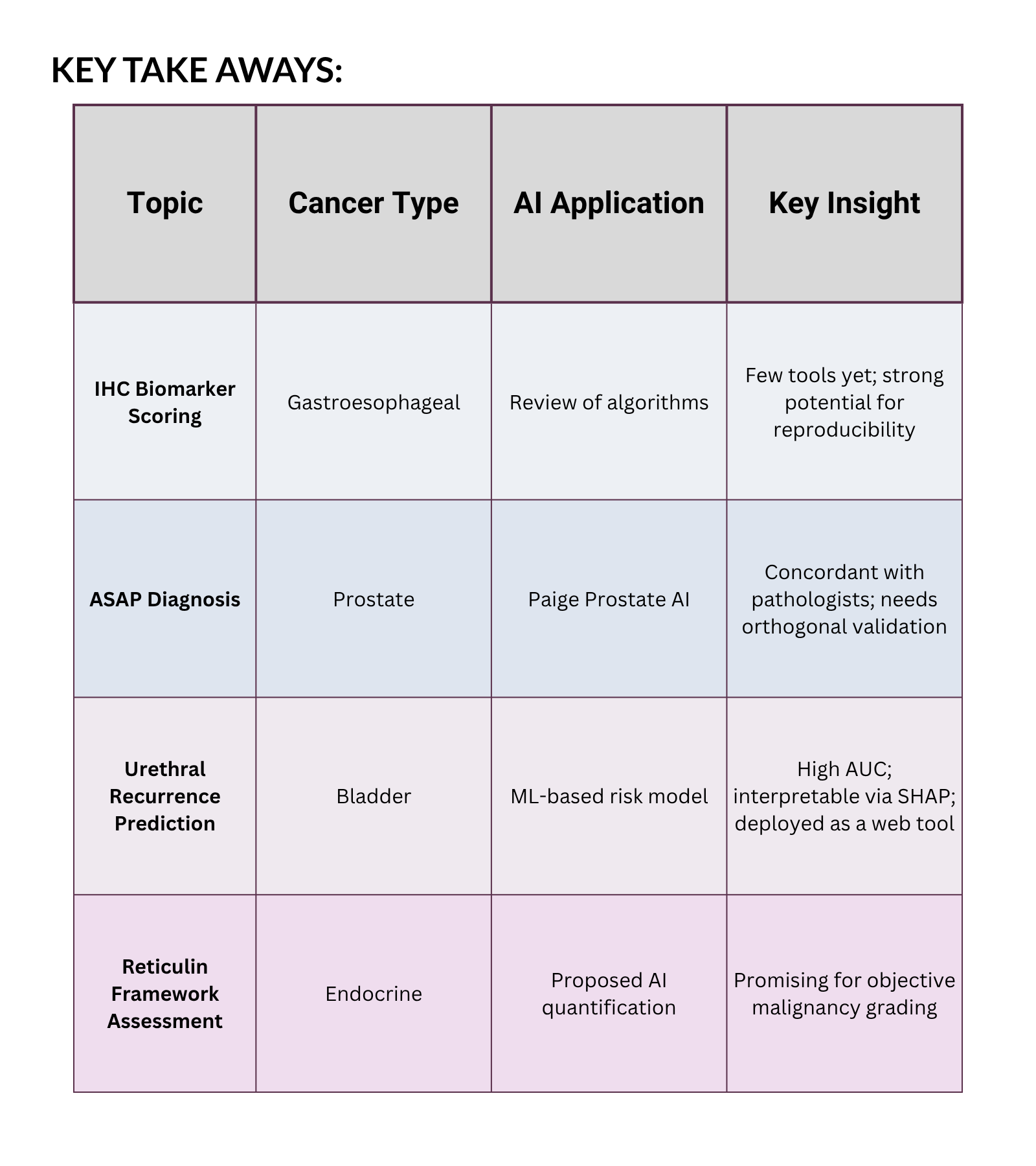
But once we got rolling, things got really interesting. This episode was all about how AI is making its way into some of the trickiest corners of pathology—from immunohistochemical (IHC) scoring in gastroesophageal cancers to refining ambiguous prostate biopsy diagnoses, predicting recurrence in bladder cancer, and even rethinking how we assess the reticulin framework in endocrine tumors.
And yes, before we dived into all that science, I showed off my new histology-inspired earrings—a multinucleated giant cell on one side and cartilage on the other. Because why not start the morning with a little tissue fashion?
Back to the Basics: A New Season of AI Paper Reviews
After wrapping up our seven-part AI series, this was a return to our regular rhythm—reviewing new and relevant studies that shape how we think about digital and computational pathology.
But before the science talk, a quick personal update:
I just came back from an online creator conference in Idaho (yes, virtual, but still worth it). I’m also preparing for the upcoming Society of Toxicologic Pathology (STP) meeting in Chicago, and planning some histology live streams this summer—focused on tissue recognition for beginners.
I also shared something exciting for our veterinary friends: Muse, one of our supporters, now offers digital pathology tissue imaging for veterinary medicine. That’s a big step forward for our furry and feathered patients. 🐕🐈
Now, onto the good stuff—AI, algorithms, and the future of diagnostic precision.
1️⃣ AI in IHC Biomarker Scoring for Gastroesophageal Cancer
Let’s start with the Italian group led by Alessandro Caputo et al., who looked at how AI can help with IHC biomarker scoring in gastroesophageal adenocarcinoma and squamous cell carcinoma.
If you’ve ever tried to score PD-L1 or HER2, you know what a headache it can be. 🧠
It’s time-consuming, subjective, and prone to variability even among seasoned pathologists.
Their review found that while AI-assisted IHC scoring has gained traction in breast and lung cancers, there’s very limited application in GEC. The authors call for more development of algorithms tailored to these tumor types—because an AI trained on breast tissue won’t necessarily know what to make of gastric mucosa.
My take:
This is one of those areas where AI could shine. The biggest advantage isn’t replacing us—it’s making our work more reproducible. AI can provide consistent, quantitative metrics for staining intensity and distribution, freeing us from the “guestimation” trap.
It’s not about losing our judgment; it’s about using it better—to interpret, not to measure pixels.
2️⃣ AI in Diagnosing Atypical Small Acinar Proliferation (ASAP) in Prostate Core Biopsies
Ah, ASAP—the diagnosis that keeps everyone up at night. It sits uncomfortably between benign and malignant, forcing pathologists (and urologists) to navigate ambiguity.
A recent study used Paige Prostate, one of the few FDA-approved AI pathology systems, to analyze 107 core biopsies initially diagnosed as ASAP.
Here’s what they found:
- Two pathologists reclassified the cores as benign or malignant, with 85% agreement.
- Paige Prostate classified cores as suspicious or not suspicious and achieved 66–75% concordance with the pathologists.
- Interestingly, the pathologists agreed with each other 77% of the time—so the AI wasn’t far off.
So, what’s the verdict?
Performance-wise, Paige did well. But is it useful in ASAP cases? The answer isn’t clear yet. The host (me!) raised a question that still bugs me:
👉 Did they validate the AI predictions with immunohistochemistry?
Because while the system’s numbers are impressive, ASAP cases are exactly where secondary validation (e.g., AMACR, p63, or basal cell markers) can make the difference between “watch and wait” and “start treatment.”
AI might be able to flag uncertainty—but it still needs our clinical context to close the loop.
3️⃣ Machine Learning Model to Predict Urethral Recurrence After Radical Cystectomy
Next, we moved to bladder cancer—specifically, predicting urethral recurrence after radical cystectomy.
A Chinese research team built a machine learning model using data from 4,730 patients (2010–2020). They wanted to create a practical tool for clinicians, not just another algorithm buried in a paper.
Here’s how it worked:
- Data split: 80% training, 20% testing
- Metrics used: AUC, F1 score, sensitivity, accuracy, C-index, and Brier score
- The final model achieved an AUC of 0.86 (training) and 0.77 (test)—strong, especially for clinical data.
- Predictive factors included tumor location, multifocality, bladder neck involvement, lymphovascular invasion, and adjuvant therapy type.
The team even deployed an online risk calculator—so clinicians can enter patient data and get real-time risk scores.
And to make it interpretable, they used SHAP (Shapley Additive Explanations) to visualize which features contributed most to the prediction.
My thoughts:
This is the direction I love seeing—AI tools that are usable and transparent. SHAP plots make the model’s logic understandable, which builds trust.
Imagine if we integrate this with large language models one day—where a clinician could just say, “What’s this patient’s recurrence risk?” and get both the number and the reasoning.
That’s AI meeting medicine halfway.
4️⃣ The Reticulin Framework in Endocrine Pathology – A Hidden AI Opportunity
Now, here’s the part that got everyone in the chat buzzing.
Let’s talk about reticulin—yes, that fine black mesh we see in silver stains, composed mostly of type III collagen.
It’s not flashy, but it’s critical in differentiating normal from neoplastic tissue in endocrine organs: pituitary, parathyroid, adrenal, and neuroendocrine tumors.
When that meshwork breaks down, it signals loss of normal architecture, and often malignancy. Right now, this assessment is qualitative—we eyeball it.
But what if AI could quantify it?
Think about it:
- In pituitary neuroendocrine tumors, reticulin loss distinguishes hyperplasia from adenoma.
- In parathyroid lesions, the pattern varies by tumor type.
- In adrenal cortex, it helps grade malignancy.
- And in neuroendocrine tumors, more disruption correlates with higher grade.
We could train a weakly supervised model on reticulin-stained images to quantify architectural integrity, correlating with tumor grade and outcome.
That’s not just automation—that’s augmentation.
And no one’s really doing it yet.
So if you’re a researcher looking for your next project… consider this my formal pitch.
When Histology Meets Jewelry – and Why It Matters
If you’ve been following me for a while, you know I like to mix science with creativity. So yes—those histology earrings are real, and they’re now available in the Digital Pathology Place online store (limited edition: 25 per pattern).
They’re meant to spark curiosity—like, “Why do you have cartilage hanging from your ear?”
And that’s the point. Science deserves to be talked about outside of conferences and labs. It’s a conversation starter, and sometimes that’s all it takes to inspire someone to learn more.
Big Picture: Where AI Fits in Our Pathology Workflow
Across these four papers, one pattern keeps emerging:
AI is not taking over pathology—it’s becoming the scaffolding that supports us, much like that reticulin network.
- It brings reproducibility to IHC scoring.
- It adds clarity to ambiguous diagnoses.
- It offers predictive foresight for clinical outcomes.
- It opens new quantitative doors in morphology.
But it also reminds us to stay vigilant.
Tools like Perplexity and GPT sometimes “hallucinate” citations or make up data. We need to keep verifying, questioning, and interpreting—because that’s what science is.
Final Thoughts
This livestream reminded me why I love this field. It’s the mix of precision, curiosity, and creativity.
We’re not just labeling slides anymore—we’re designing smarter workflows.
We’re not fighting AI—we’re training it to think with us.
So, whether you joined live at sunrise or are catching up later with your coffee, thank you for being part of this journey.
And if you’re a pathology researcher or trainee—remember: every stain, every pixel, every odd artifact could be the key to the next breakthrough.
See you at the next DigiPath Digest, and until then—stay curious, stay caffeinated, and keep looking closer.













Comments are closed.