You’ve invested in a state-of-the-art whole slide scanner. Your lab has cutting-edge AI algorithms ready to deploy. But there’s a problem: they can’t talk to each other.
The scanner outputs images in a proprietary format. The AI tool requires yet another format. Your image management system speaks a different language entirely. And now you’re spending weeks—or months—trying to build bridges between systems that should have been designed to work together from the start.
If this sounds familiar, you’ve encountered the fundamental challenge holding back digital pathology: the lack of interoperability. This is where open pathology comes in.
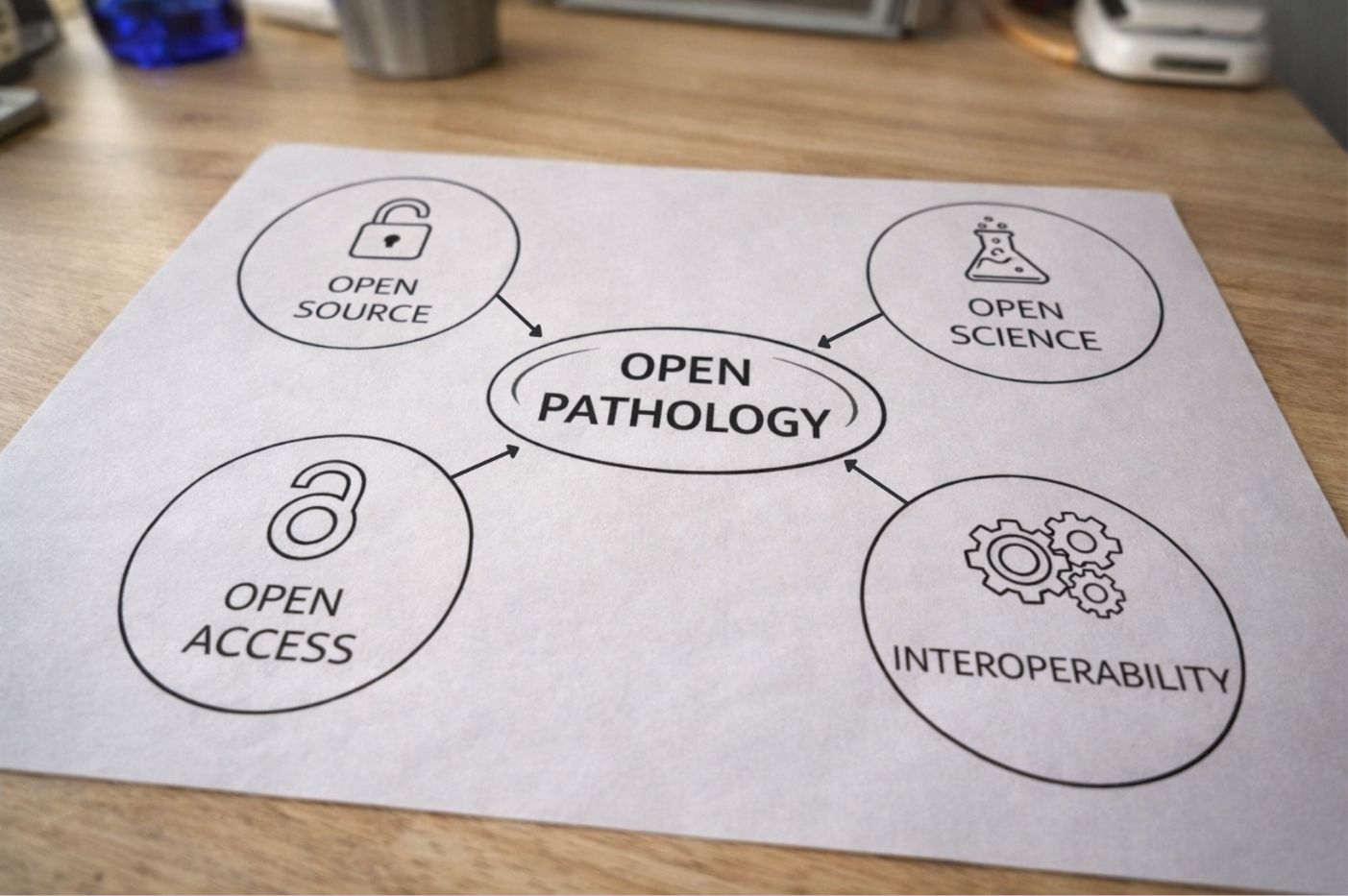
Diagram illustrating how open pathology builds on established open science and open source principles
The concept of “open” isn’t new in science and technology. Open-source software powers most of the internet. Open-access publishing has transformed scientific communication. Open science initiatives have accelerated research by enabling data sharing across institutions.
Open pathology applies these same principles to digital pathology systems. At its core, open pathology means building digital pathology ecosystems where different vendors’ tools can seamlessly exchange data, where labs maintain control and flexibility in their technology choices, and where innovation isn’t constrained by proprietary barriers.
This approach isn’t just theoretical. We’ve seen it work in radiology.
What Radiology Learned the Hard Way
In the early days of digital radiology, each equipment manufacturer used proprietary image formats. CT scanners from different vendors produced images that couldn’t be easily shared or compared. Radiologists needed multiple viewing systems. Hospitals faced vendor lock-in, unable to upgrade systems without replacing their entire infrastructure.
According to research on the DICOM standard’s development, the American College of Radiology and the National Electrical Manufacturers Association recognized this problem in the early 1980s. By 1985, they released the first ACR-NEMA standard for digital imaging. This eventually evolved into DICOM (Digital Imaging and Communications in Medicine), which became the universal standard for medical imaging.
The transformation took time—nearly a decade before widespread adoption—but the results speak for themselves. Today, DICOM enables any radiology device to communicate with any PACS (Picture Archiving and Communication System), regardless of manufacturer. This standardization didn’t stifle innovation; it accelerated it by allowing developers to focus on improving capabilities rather than building proprietary data silos.
Digital pathology now faces the same inflection point radiology encountered thirty years ago.
The Hidden Costs of Closed Systems
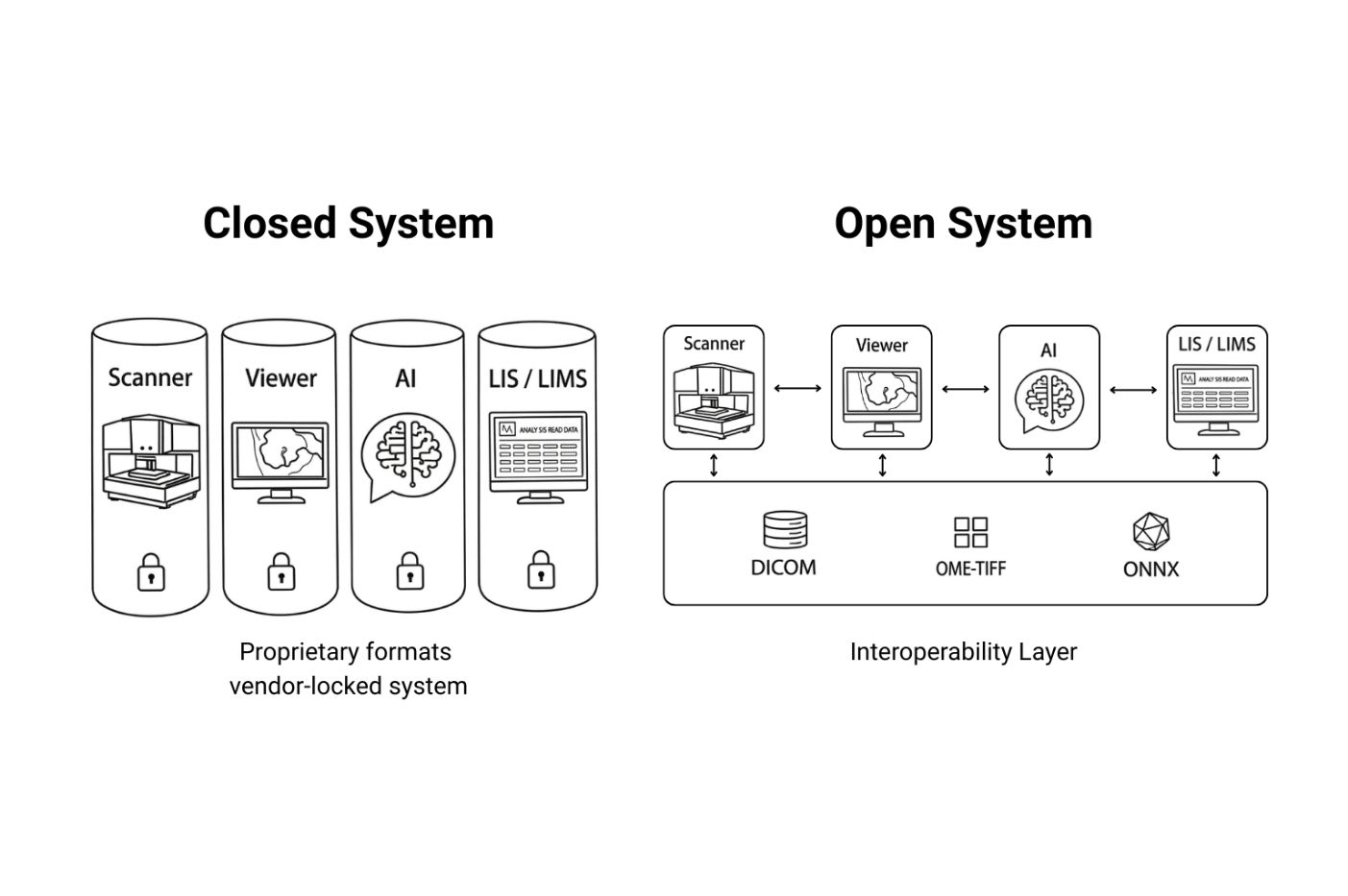
Comparison showing closed digital pathology systems with isolated components versus open systems with interoperable components
When digital pathology systems can’t interoperate, labs face challenges that go beyond technical frustration:
Friction in the diagnostic workflow: Pathologists who must log into separate systems, navigate different interfaces, and toggle between applications spend less time on diagnosis. According to research on digital pathology workflows, even small workflow interruptions accumulate, affecting both diagnostic quality and pathologist satisfaction.
Barriers to AI adoption: Labs often have existing digital pathology infrastructure. When an FDA-cleared AI algorithm becomes available, deployment depends entirely on whether it can integrate with current systems. Closed ecosystems force labs to choose between adopting beneficial AI tools and maintaining their existing workflow, effectively limiting patient access to diagnostic innovations.
Limited vendor options: When a lab’s image management system only works with specific scanners, or when AI algorithms can only be deployed on proprietary platforms, the lab loses negotiating power. They’re locked into a single vendor’s ecosystem, unable to adopt better or more cost-effective solutions as they become available.
Adaptation challenges: Every lab operates differently, shaped by its patient population, subspecialty focus, and organizational structure. Rigid, closed systems that can’t be adapted to specific workflows force labs to change their processes to fit the software, rather than configuring software to support optimal diagnostic workflows.
Future-proofing concerns: Technology evolves rapidly. Pathology labs making infrastructure investments today need systems that will remain compatible with future innovations. Closed systems create uncertainty about whether current investments will support or constrain future capabilities.
The Building Blocks of Open Pathology
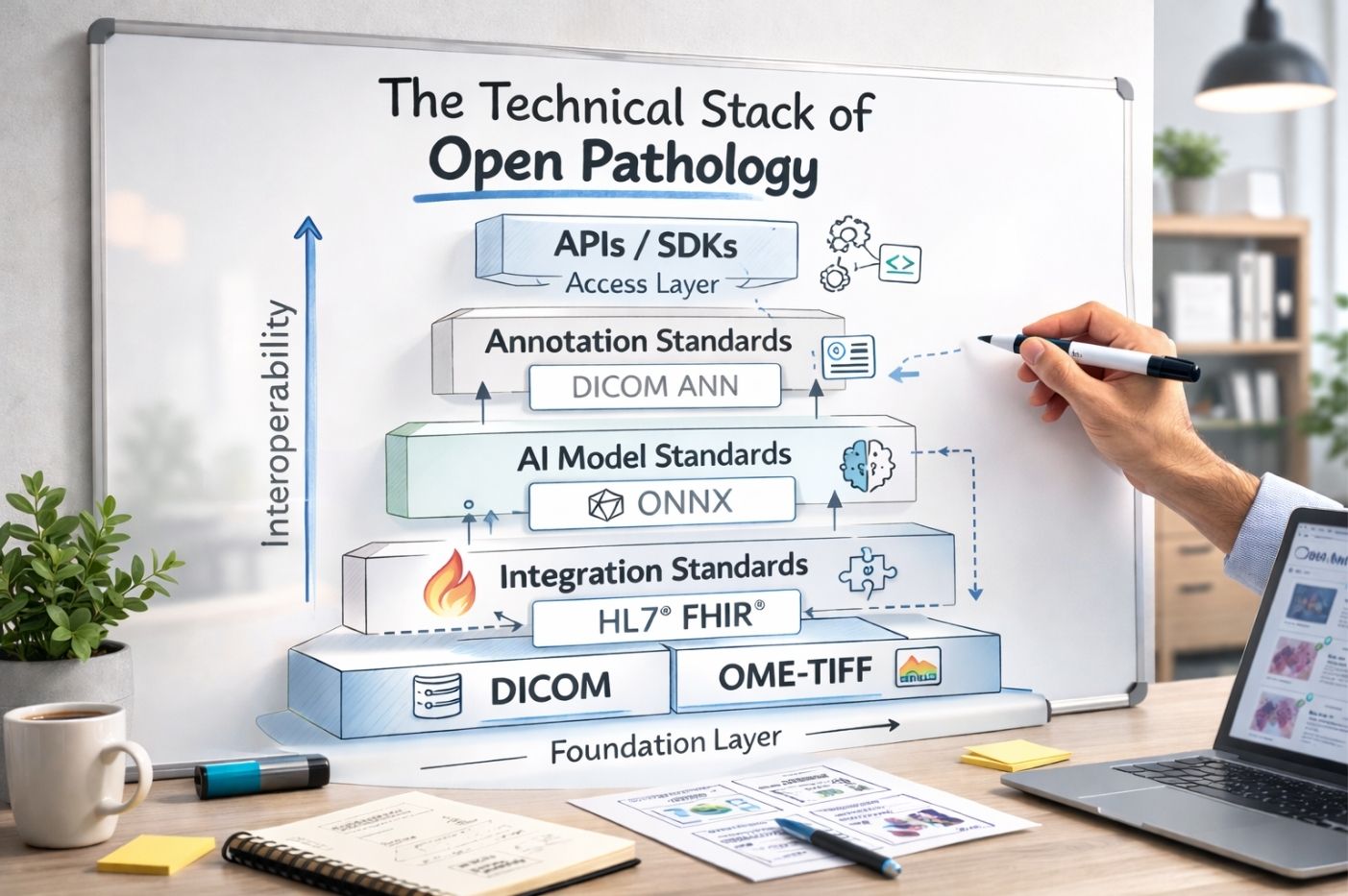
Infographic illustrating the technical layers that enable open pathology interoperability
Open pathology isn’t just about good intentions—it requires concrete technical foundations. Several key components enable true interoperability:
Open Image Standards
The pathology field has developed open standards for storing and exchanging whole slide images. DICOM, the same standard that transformed radiology, has been extended to support pathology through the work of DICOM Working Group 26 (WG-26), established in 2005 specifically for pathology and whole slide imaging.
OME-TIFF (Open Microscopy Environment TIFF) provides another open standard, widely supported across research applications. When digital pathology platforms support these open standards, images can move freely between systems without requiring proprietary conversion tools.
AI Model Portability
For artificial intelligence to reach its potential in pathology, trained models need to move between platforms. The ONNX (Open Neural Network Exchange) format enables this portability. When platforms support ONNX, labs can train AI models in one environment and deploy them in another, or import FDA-cleared models developed by third parties without being constrained by platform compatibility.
Annotation Interoperability
According to DICOM WG-26’s recent work, standardized annotation formats are essential for both manual pathologist annotations and AI-generated analysis results. The DICOM Annotation (ANN) standard, introduced in 2021, allows efficient storage and retrieval of bulk annotation data, supporting both manual and AI-generated annotations. This standardization enables pathologists to annotate regions of interest and view AI analysis results without leaving their primary viewing platform.
Laboratory Information System Integration
Digital pathology doesn’t exist in isolation—it’s part of the broader laboratory workflow. Open pathology platforms support standards like HL7 and HL7 FHIR for integration with laboratory information systems (LIS) and electronic health records (EHR). This integration ensures that pathology images and analysis results flow seamlessly into the complete patient record.
Open APIs and Development Tools
Well-documented, open application programming interfaces (APIs) enable custom integrations. When vendors provide software development kits (SDKs), labs with specific needs can build custom modules or integrations tailored to their workflows. This extensibility transforms a platform from a fixed product into an adaptable foundation.
The Building Blocks of Open Pathology
Open pathology requires collaboration. When vendors commit to open standards, they’re committing to work with—not against—other companies in the ecosystem.
Look at how digital pathology workflows actually function: a whole slide scanner from one vendor needs to send images to an image management system from another vendor, which then connects to AI algorithms from multiple developers, while integrating with the lab’s LIS. Each of these integrations requires technical coordination and ongoing support.
Companies that embrace open pathology proactively establish partnerships with complementary vendors. These partnerships benefit labs by reducing the integration burden—instead of each lab negotiating separate agreements and troubleshooting compatibility issues, participating vendors handle these technical details in advance.
The presence of established partnerships serves as a practical indicator of a vendor’s commitment to openness. When companies work together to ensure their products interoperate smoothly, labs gain confidence that their technology investments will work together rather than creating new silos.
Case Study: Indica Labs – Built on Open Pathology Principles
Open pathology requires collaboration. When vendors commit to open standards, they’re committing to work with—not against—other companies in the ecosystem.
Look at how digital pathology workflows actually function: a whole slide scanner from one vendor needs to send images to an image management system from another vendor, which then connects to AI algorithms from multiple developers, while integrating with the lab’s LIS. Each of these integrations requires technical coordination and ongoing support.
Companies that embrace open pathology proactively establish partnerships with complementary vendors. These partnerships benefit labs by reducing the integration burden—instead of each lab negotiating separate agreements and troubleshooting compatibility issues, participating vendors handle these technical details in advance.
The presence of established partnerships serves as a practical indicator of a vendor’s commitment to openness. When companies work together to ensure their products interoperate smoothly, labs gain confidence that their technology investments will work together rather than creating new silos.
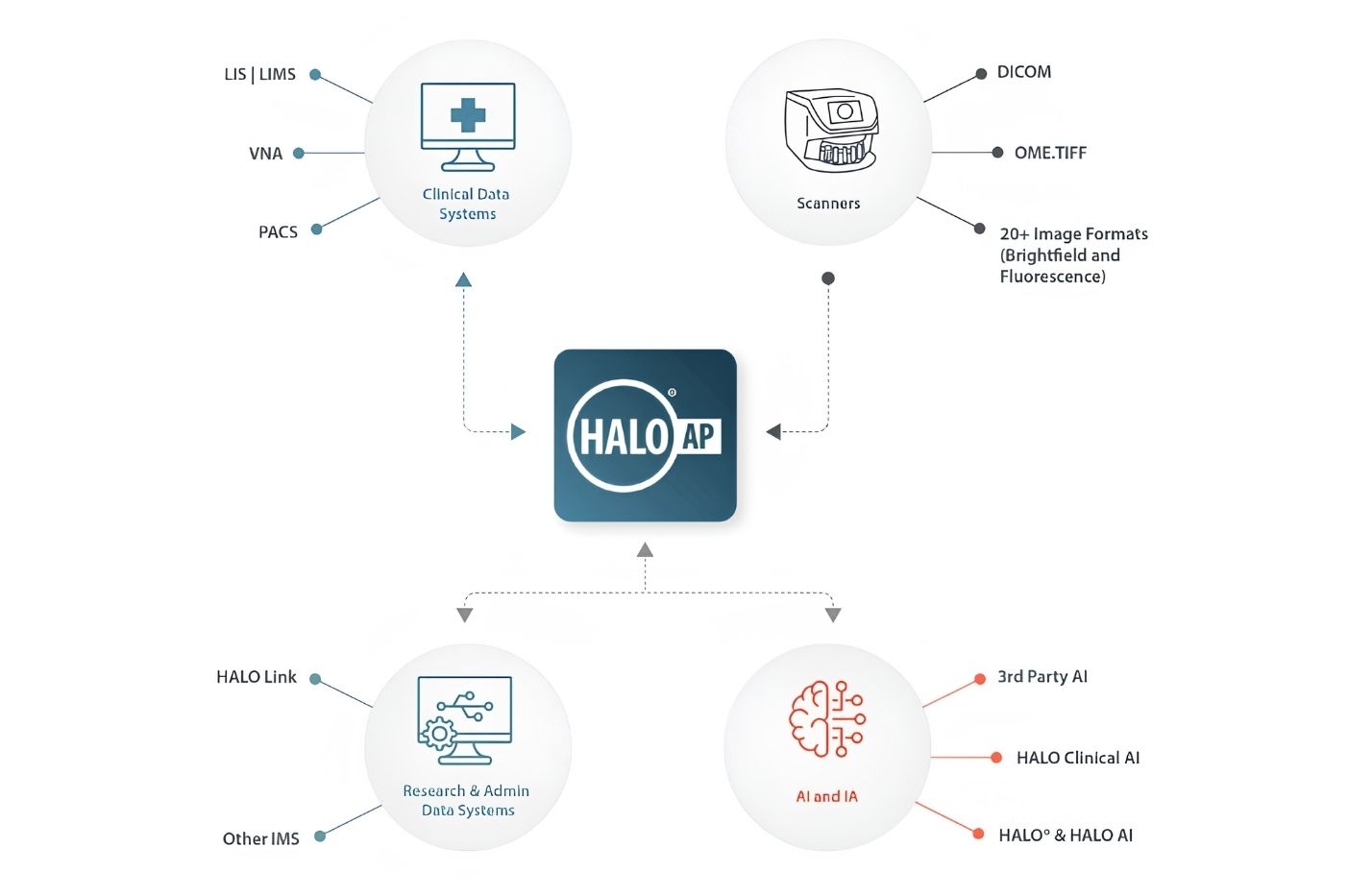
HALO AP digital pathology platform interface showing integration of third-party AI analysis
Indica Labs has demonstrated commitment to open pathology throughout development of their HALO platforms, both for life sciences research and clinical applications.
Standards Support: All HALO platforms support open image standards including OME and DICOM formats for images. For AI models, HALO AI supports the ONNX format, enabling users to both import external models and export trained models. For LIS and EHR integration, HALO platforms support HL7 and HL7 FHIR standards.
Integration-First Design: An integration-focused architecture underpins HALO platforms, which are built with open, well-defined APIs supported by comprehensive documentation and sample code. For users requiring specialized functionality, software development kits enable custom module creation tailored to specific workflows.
Scanner Agnostic Approach: HALO platforms support image files from all leading scanner manufacturers, allowing labs to choose scanners based on their specific needs rather than compatibility requirements. Indica Labs actively collaborates with scanner developers to ensure ongoing compatibility.
AI Integration Partnerships: Consistent with open pathology principles, Indica Labs has partnered with leading AI developers to integrate third-party solutions with HALO platforms. Current partnerships include integrations with Lunit SCOPE PD-L1, Paige (now a Tempus company) Diagnostic AI models, IBEX models, Histofy, DeepBio and Visiopharm’s Discovery. These integrations allow labs to access best-of-breed AI tools alongside HALO platforms’ own AI capabilities, enabling seamless result viewing directly within HALO interfaces.
This approach empowers labs to build customized, scalable digital pathology ecosystems that meet their specific needs while remaining flexible as new technologies emerge.
The Path Forward
Digital pathology stands at a critical juncture. Labs investing in digital infrastructure today are making decisions that will shape their capabilities for the next decade. The choice between closed, proprietary systems and open, interoperable platforms will determine not just technical flexibility, but ultimately how quickly diagnostic innovations reach patients.
Open pathology isn’t about choosing a specific vendor or technology—it’s about insisting on systems built for collaboration rather than isolation. It’s about demanding that digital pathology software serve the needs of pathologists and patients, not the business models of individual vendors.
The technical foundations exist. The standards are established. What remains is for labs to prioritize interoperability when evaluating digital pathology platforms, and for vendors to compete on capabilities and service rather than on their ability to lock customers into proprietary ecosystems.
When pathology follows radiology’s path toward standardization, the entire field will benefit from accelerated innovation, reduced costs, and most importantly, improved patient care through broader access to diagnostic tools.
Interested in learning how open pathology principles can benefit your lab? Contact Indica Labs at info@indicalab.com to discuss how the HALO platforms can support your specific workflow needs while maintaining flexibility for future innovation.
Regulatory Note: HALO AP® is CE-IVDR marked for in-vitro diagnostic use in Europe, the UK, and Switzerland. HALO AP® is For Research Use Only in the USA and is not FDA cleared for clinical diagnostic use.












Comments are closed.